Neuro degenerative diseases
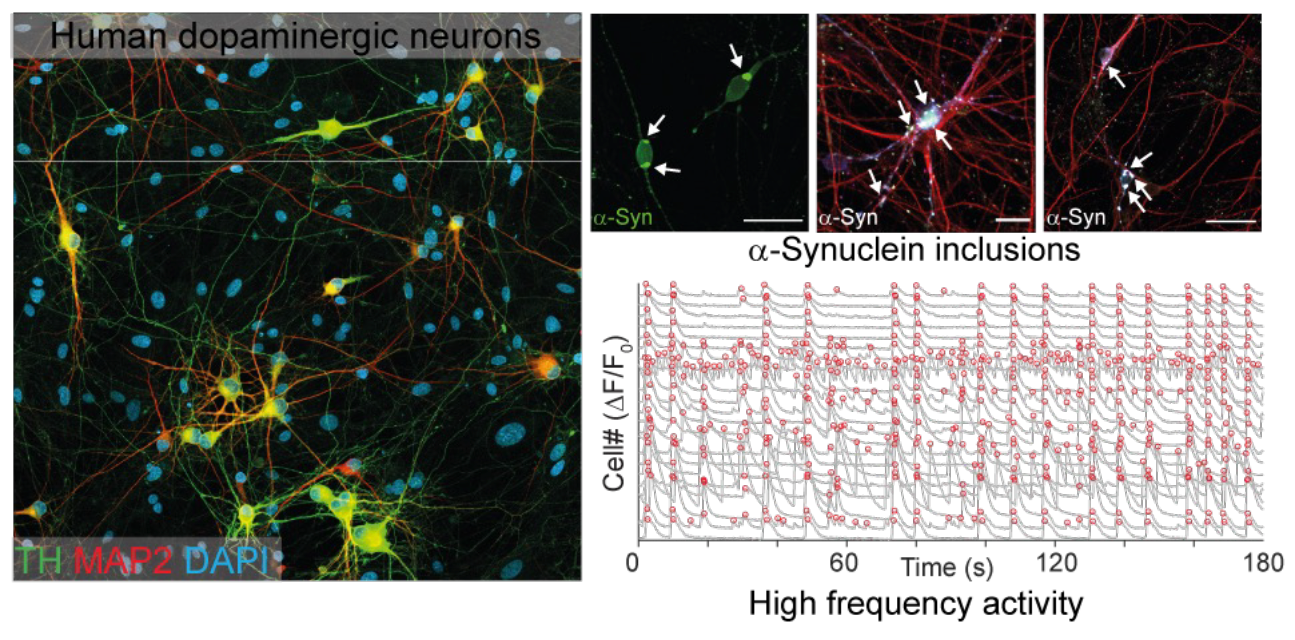
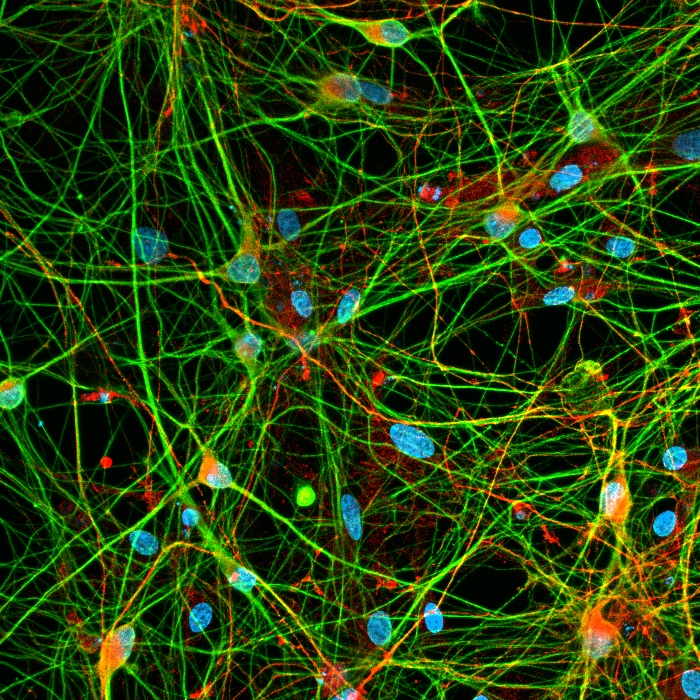
We generate human iPSC-derived dopaminergic neurons using transcription factor–based reprogramming combined with ventralizing patterning factors to produce highly pure and reproducible cultures. Our optimized differentiation protocol consistently yields >95% tyrosine hydroxylase (TH)-positive neurons that exhibit hallmark features of midbrain dopaminergic neurons, including spontaneous pacemaking activity, synaptic connectivity, and dopamine-related neuronal physiology.
These cultures are compatible with high-content functional assays and scalable screening approaches, enabling quantitative assessment of neuronal dysfunction and therapeutic rescue in Parkinson’s disease-relevant systems.
Parkinson’s Disease Phenotypic Assays
Our platform enables multiparametric characterization of Parkinson’s disease phenotypes, including:
- spontaneous pacemaking activity and neuronal excitability
- neuronal network activity and synaptic transmission
- dopaminergic neuron survival and degeneration kinetics
- neurite outgrowth and degeneration
- mitochondrial dynamics and organelle transport
- lysosomal function and autophagy pathways
- α-synuclein aggregation and inclusion formation
- cellular stress responses and vulnerability to toxic stimuli
These assays allow quantitative evaluation of disease-associated phenotypes, target engagement, and functional rescue following therapeutic intervention.
Available IPSC disease models
- Extensively characterized idiopathic and genetic Parkison’s disease iPSC lines.
- Gene-edited models with disease mutations and matched isogenic-controls
- Highly scalable dopaminergic cultures that spontaneously develop ⍺-Synuclein inclusions resembling those observed in post-mortem Parkinson’s disease brain tissue.
These models provide a powerful system to study genetic risk factors, pathogenic mechanisms, and compound efficacy in human dopaminergic neurons.
Importantly, our dopaminergic cultures develop α-synuclein inclusions without artificial overexpression, closely recapitulating pathology observed in Parkinson’s disease brain tissue
Model characterization
All models undergo extensive molecular, morphological, and functional validation, including:
- neuronal network activity measurements
- spontaneous pacemaking electrophysiology
- neurite growth and degeneration assays
- mitochondrial and organelle transport dynamics
- α-synuclein aggregation and pathology assays
- synaptic connectivity and neurotransmission
This deep phenotypic characterization enables robust benchmarking of disease phenotypes and quantitative assessment of therapeutic rescue.
Human dopaminergic neurons